Framework for the pharmacological treatment of obesity and its complications from the European Association for the Study of Obesity (EASO): 2026 update.
https://doi.org/10.1038/s41591-026-04397-4
The updated EASO framework helps clinicians match pharmacological obesity management to an individual’s health needs, moving beyond weight loss alone to consider complications, mechanisms and treatment goals.


Download this Infographic in PDF Format.
We were pleased to launch EASO Algorithm 2.0 at ECO2026 this week. The 2026 update to the EASO pharmacological framework for obesity treatment reflects the rapidly evolving evidence base for obesity management medications, particularly for incretin-based therapies. The framework provides a practical, evidence-informed algorithm to support clinical decision-making.
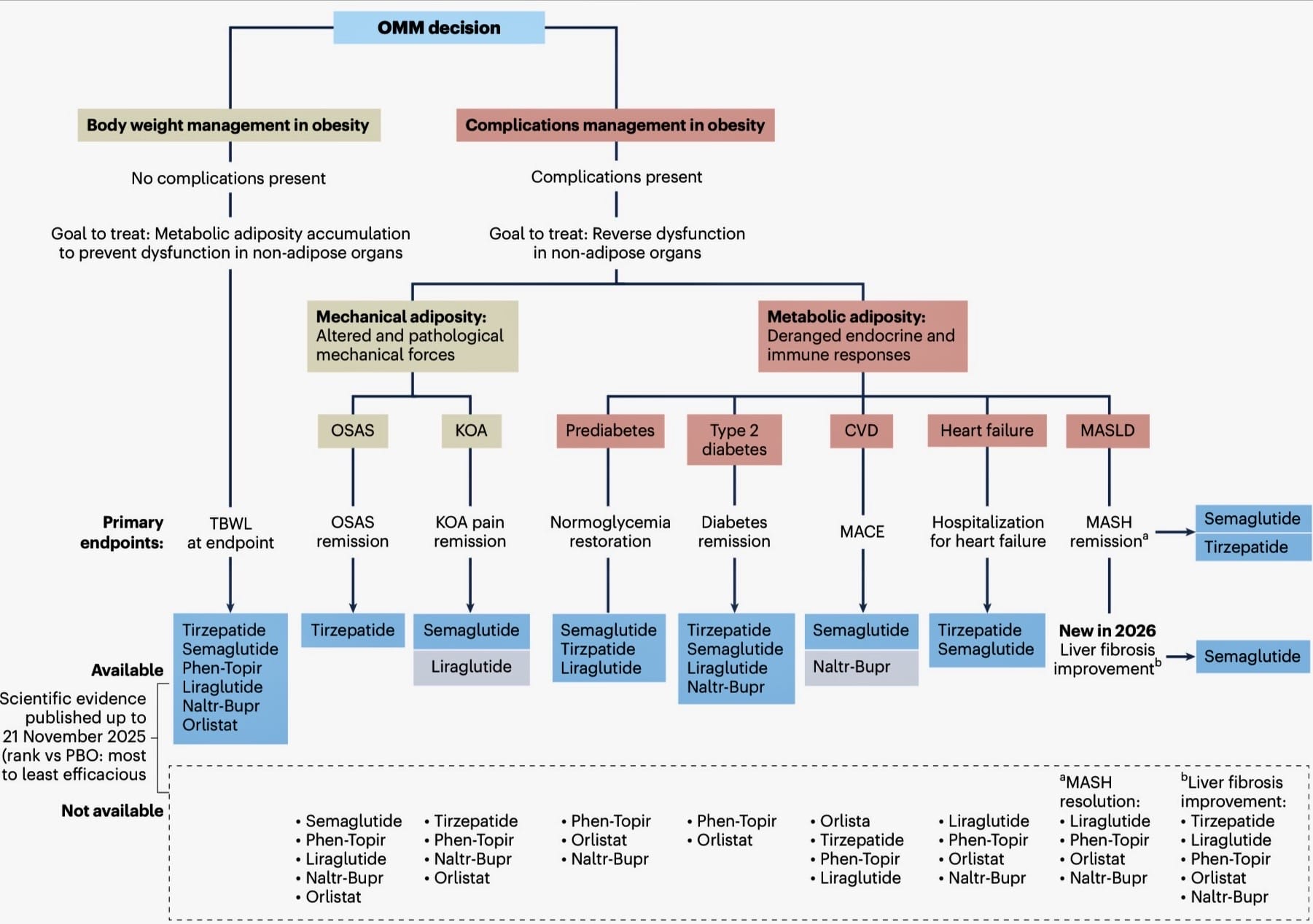
The updated framework, which uses the original published Algorithm methodology, is based on 62 randomised controlled trials, incorporating evidence published up to 21 November 2025. It refines the 2025 EASO algorithm, strengthening evidence around total body weight loss, clarifying the comparative parameters of available medications, and expanding the liver disease domain to include MASH resolution and, newly for 2026, liver fibrosis improvement.
The central message within the algorithm is that pharmacological obesity management should be personalised. Treatment decisions should consider not only body weight outcomes, but also the presence and severity of obesity-related complications, including type 2 diabetes, cardiovascular diseases, heart failure, obstructive sleep apnoea, knee osteoarthritis and MASLD/MASH.
Updates from the 2.0 framework
The 2026 update confirms EASO’s intention to maintain the pharmacological framework as a living evidence map, updated as new trials, outcomes, medications and formulations emerge.
Evidence for body weight management
For total body weight loss, all medications assessed showed clinically meaningful benefit versus placebo. The strongest weight-loss effects were seen with tirzepatide and semaglutide, with tirzepatide presenting above semaglutide in the body weight management domain.
Strong focus on obesity-related complications
The framework distinguishes between:
- Body weight management in obesity, where no complications are present and the intention of treatment is to prevent dysfunction in non-adipose organs and
- Complications management in obesity, where complications are already present and the goal is to reverse or improve dysfunction in non-adipose organs.
New liver disease evidence emerges
The most substantive 2026 update is in the liver domain. The framework now includes:
- MASH resolution, where current evidence supports benefit for both semaglutide and tirzepatide.
- Liver fibrosis improvement, newly added in 2026, where current evidence is strongest for semaglutide, while evidence for tirzepatide is less mature.
Mechanism-informed care
The framework supports a more comprehensive and precise understanding of obesity through two concepts:
- Metabolic adiposity: linked to deranged endocrine and immune responses, contributing to complications such as prediabetes, type 2 diabetes, cardiovascular diseases, heart failure and MASLD/MASH.
- Mechanical adiposity: linked to altered physical forces, contributing to complications such as obstructive sleep apnoea and knee osteoarthritis.
Congratulations to the authors. Read the publication: https://doi.org/10.1038/s41591-026-04397-4
